News Story
Dimitrakopoulos Wins NBIB/NIH Grant for Study on Disease States of Red Blood Cells

Dimitrakopoulos is the director of the BioFluid Dynamics Laboratory, which applies novel computational and theoretical methodologies for the accurate and efficient study of the dynamics of drops and bubbles in microfluidics and porous media, hemodynamics and hemopathology in the microcirculation, and the dynamics of synthetic and biological polymers.
ChBE Professor Panos Dimitrakopoulos was recently awarded a grant on "Disease States of Red Blood Cells in Vascular Microvessels" from the National Institute of Biomedical Imaging and Bioengineering (NIBIB), part of the National Institutes of Health (NIH). The research will be conducted in Dimitrakopoulos' Biofluid Dynamics Computational Laboratory, and involves study of the effects of paraproteinemia and malaria on the motion of the erythrocytes in the microcirculation. Paraproteinemia is a condition associated with elevated plasma protein levels. Common disorders associated with paraproteinemia are rheumatoid arthritis (a chronic inflammatory disease characterized by joint swelling and pain), Cushing syndrome and diabetes mellitus. Malaria is a disease which affects nearly 8% of the world population, causing 2 to 3 million deaths every year.
 |
|
|
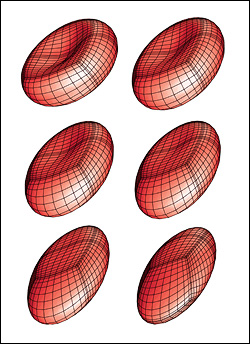
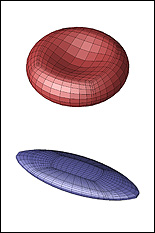
A biconcave erythrocyte in a strong shear flow obtains an inclined ellipsoidal conformation while its membrane tank-treads around the cell. |
|
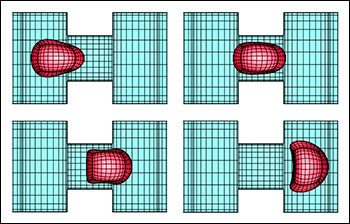
Both abnormal physiological states affect the ability of red blood cells to deform and thus to pass through the networks of microvessels. This lowers the amount of oxygen delivered to the body's tissues with mild or severe health consequences. Unfortunately, in vivo measurements of the blood resistance in microvascular segments are scarce; these measurements require simultaneous insertion of two micropipettes in a vessel segment and are technically very difficult. The proposed computational investigation of realistic problems in the microcirculation is a very attractive alternative. In order to accurately describe the motion of affected erythrocytes in blood capillaries, Dimitrakopoulos' laboratory will employ an advanced spectrally-accurate computational algorithm that they recently developed under a National Science Foundation grant. (See related story.) This Spectral Boundary Element algorithm facilitates considerably the study of the complicated three-dimensional erythrocyte dynamics in the microcirculation, such as cells in close contact, cells in proximity to the walls of the blood capillaries, and large cell deformations. "For both types of abnormalities, our computational investigation will elucidate the deformation and motion of erythrocytes and the associated resistance in blood flow", Dimitrakopoulos explains. "Of particular interest is the variation of hematocrit and the changes in the blood apparent viscosity which occurs in these vessels due to the strong hydrodynamic interactions of the cells with the vessel walls. Our work will also help elucidate the erythrocytes' aging and lifetime since the large deformations and the associated large interfacial stresses may damage the cell's membrane and accelerate the aging process. We hope that the proposed work will advance our understanding of physiological systems, improve the control of these diseases and thus enhance public health, in agreement with the goals of NIH." To learn more about Professor Dimitrakopoulos' work and research group, visit www.eng.umd.edu/~dimitrak/
Published May 4, 2009